Creatine
| Creatine | |
|---|---|
 |
 |
|
2-(methylguanidino) ethanoic acid
|
|
|
Other names
• (α-Methylguanido)acetic acid
• Creatin • Kreatin |
|
| Identifiers | |
| CAS number | 57-00-1 |
| PubChem | 586 |
| ChemSpider | 566 |
| EC number | 200-306-6 |
|
SMILES
O=C(O)CN(C(=[N@H])N)C
|
|
|
InChI
InChI=1/C4H9N3O2/c1-7(4(5)6)2-3(8)9/h2H2,1H3,(H3,5,6)(H,8,9)
Key: CVSVTCORWBXHQV-UHFFFAOYAV |
|
| Properties | |
| Molecular formula | C4H9N3O2 |
| Molar mass | 131.13 g mol−1 |
| Melting point |
303 °C (decomp.) |
| Except where noted otherwise, data are given for materials in their standard state (at 25 °C, 100 kPa) |
|
| Infobox references | |
Creatine is a nitrogenous organic acid that occurs naturally in vertebrates and helps to supply energy to muscle. Creatine was identified in 1832 when Michel Eugène Chevreul discovered it as a component of skeletal muscle, which he later named creatine after the Greek word for flesh, κρέας (kreas).
Contents |
Biosynthesis
Creatine is naturally produced in the human body from amino acids primarily in the kidney and liver. It is transported in the blood for use by muscles. Approximately 95% of the human body's total creatine is located in skeletal muscle.[1]
Creatine is not an essential nutrient, as it is manufactured in the human body from L-arginine, glycine, and L-methionine.[2]
In humans and animals, approximately half of stored creatine originates from food (mainly from meat). Since vegetables do not contain creatine, vegetarians show lower levels of muscle creatine, but show the same levels after using supplements.[3]
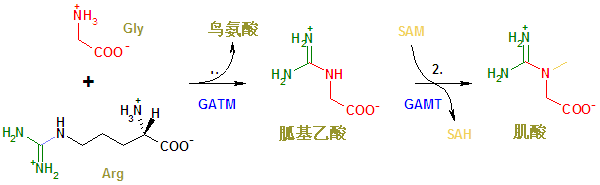
Arg - Arginine; GATM - Glycine amidinotransferase; GAMT - Guanidinoacetate N-methyltransferase; Gly - Glycine; Met - Methionine; SAH - S-adenosyl homocysteine; SAM - S-adenosyl methionine.
The enzyme GATM (L-arginine:glycine amidinotransferase (AGAT), EC 2.1.4.1) is a mitochondrial enzyme responsible for catalyzing the first rate-limiting step of creatine biosynthesis, and is primarily expressed in the kidneys and pancreas.[4]
The second enzyme in the pathway (GAMT, guanidinoacetate N-methyltransferase, EC:2.1.1.2) is primarily expressed in the liver and pancreas.[4]
Genetic deficiencies in the creatine biosynthetic pathway lead to various severe neurological defects.[5]
Health effects
Use as food supplement
Creatine supplements are sometimes used by athletes, bodybuilders, and others who wish to gain muscle mass, typically consuming 2 to 3 times the amount that could be obtained from a very-high-protein diet. A survey of long-time use gives the creatine content of several foods.[6] The Mayo Clinic states that creatine has been associated with asthmatic symptoms and warns against consumption by persons with known allergies.[7]
While there was once some concern that creatine supplementation could affect hydration status and heat tolerance and lead to muscle cramping, recent studies have shown these concerns to be unfounded.[8][8][9][9]
There is less concern today than there used to be about possible kidney damage from creatine, although there are reports of kidney damage, such as interstitial nephritis; patients with kidney disease should avoid use of this supplement.[7] In similar manner, liver function may be altered, and caution is advised in those with underlying liver disease although studies have shown little or no adverse impact on kidney or liver function from oral creatine supplementation.[10]
In theory, creatine may alter the activities of insulin. Caution is advised in patients with diabetes or hypoglycemia, and in those taking drugs, herbs, or supplements that affect blood sugar. Serum glucose levels may need to be monitored by a health-care professional, and medication adjustments may be necessary.
Long-term administration of large quantities of creatine is reported to increase the production of formaldehyde, which has the potential to cause serious unwanted side-effects. However, this risk is largely theoretical because urinary excretion of formaldehyde, even under heavy creatine supplementation, does not exceed normal limits.[11]
Extensive research over the last decade has shown that oral creatine supplementation at a rate of 5 to 20 grams per day appears to be very safe and largely devoid of adverse side-effects [12] while at the same time, effectively improving the physiological response to resistance exercise, increasing the maximal force production of muscles in both men and women.[13][14]
Creatine has become increasingly commonly used by body builders to improve their ability to build muscle tissue. By allowing body builders to perform more work as a result of additional energy, increased protein synthesis is stimulated. Also, when an abundance of creatine phosphate is stored in the muscle, the muscle will hold more water in its cells. This process is called, "Cell Hydration", and is often confused with water retention. But, in this case, water is stored within the cell. Whereas, with "water retention" the water is outside the cell. The more water is held in a muscle cell, the more it will promote the synthesis of protein as well as deter protein breakdown.[15]
Pharmacokinetics
Endogenous serum or plasma creatine concentrations in healthy adults are normally in a range of 2–12 mg/L. A single 5 g (5000 mg) oral dose in healthy adults results in a peak plasma creatine level of approximately 120 mg/L at 1–2 hours post-ingestion. Creatine has a fairly short elimination half-life, averaging just less than 3 hours, so to maintain an elevated plasma level it would be necessary to take small oral doses every 3–6 hours throughout the day. After the "loading dose" period (1–2 weeks, 12-24g a day), it is no longer necessary to maintain a consistantly high serum level of Cr. As with most supplements, each person has their own genetic "preset" amount of creatine they can hold. The rest is eliminated out of the body as waste. Creatine is consumed by the body fairly quickly. And if one wishes to maintain the high concentration of Cr ,Post - loading dose, 2-5g daily is the standard amount to intake.[16][17][18]
Pregnancy and breastfeeding
Creatine cannot be recommended during pregnancy or breastfeeding due to a lack of scientific information. Pasteurized cow's milk contains higher levels of creatine than human milk.[19]
Treatment of diseases
Creatine has been demonstrated to cause modest increases in strength in people with a variety of neuromuscular disorders.[20] Creatine supplementation has been, and continues to be, investigated as a possible therapeutic approach for the treatment of muscular, neuromuscular, neurological and neurodegenerative diseases (arthritis, congestive heart failure, Parkinson's disease, disuse atrophy, gyrate atrophy, McArdle's disease, Huntington's disease, miscellaneous neuromuscular diseases, mitochondrial diseases, muscular dystrophy, and neuroprotection).
A study demonstrated that creatine is twice as effective as the prescription drug riluzole in extending the lives of mice with the degenerative neural disease amyotrophic lateral sclerosis (ALS, or Lou Gehrig's disease). The neuroprotective effects of creatine in the mouse model of ALS may be due either to an increased availability of energy to injured nerve cells or to a blocking of the chemical pathway that leads to cell death.[21] A similarly promising result has been obtained in prolonging the life of transgenic mice affected by Huntington's disease. Creatine treatment lessened brain atrophy and the formation of intranuclear inclusions, attenuated reductions in striatal N-acetylaspartate, and delayed the development of hyperglycemia.[22]
Cognitive ability
A placebo-controlled double-blind experiment found that vegetarians who took 5 grams of creatine per day for six weeks showed a significant improvement on two separate tests of fluid intelligence, Raven's Progressive Matrices, and the backward digit span test from the WAIS. The treatment group was able to repeat longer sequences of numbers from memory and had higher overall IQ scores than the control group. The researchers concluded that "supplementation with creatine significantly increased intelligence compared with placebo."[23] A subsequent study found that creatine supplements improved cognitive ability in the elderly.[24] A study on young adults (0.03 g/kg/day for six weeks, e.g., 2 g/day for 150-pound individual) failed, however, to find any improvements.[25]
See also
- Adenosine triphosphate
- Beta-alanine
- Creatine phosphate
References
- ↑ "Creatine". MedLine Plus Supplements. U.S. National Library of Medicine. 2010-07-20. http://www.nlm.nih.gov/medlineplus/druginfo/natural/patient-creatine.html. Retrieved 2010-08-16.
- ↑ "Creatine". Beth Israel Deaconess Medical Center. http://www.bidmc.org/YourHealth/ConditionsAZ.aspx?ChunkID=21706. Retrieved 2010-08-23.
- ↑ Burke DG, Chilibeck PD, Parise G, Candow DG, Mahoney D, Tarnopolsky M (2003). "Effect of creatine and weight training on muscle creatine and performance in vegetarians". Medicine and science in sports and exercise 35 (11): 1946–55. doi:10.1249/01.MSS.0000093614.17517.79. PMID 14600563.
- ↑ 4.0 4.1 "ETH ETH E-Collection: Methylglyoxal, creatine and mitochondrial micro-compartments - ETH E-Collection". E-collection.ethbib.ethz.ch. 2008-04-19. doi:10.3929/ethz-a-004636659.. http://e-collection.ethbib.ethz.ch/ecol-pool/diss/fulltext/eth15180.pdf. Retrieved 2010-08-16.
- ↑ "L-Arginine:Glycine Amidinotransferase". Ncbi.nlm.nih.gov. http://www.ncbi.nlm.nih.gov/entrez/dispomim.cgi?id=602360. Retrieved 2010-08-16.
- ↑ "Creatine: is it really safe for long-term use?". Pponline.co.uk. http://www.pponline.co.uk/encyc/creatine.htm. Retrieved 2010-08-16.
- ↑ 7.0 7.1 "Creatine: Safety". MayoClinic.com. http://www.mayoclinic.com/health/creatine/NS_patient-creatine/DSECTION=safety. Retrieved 2010-08-16.
- ↑ 8.0 8.1 Lopez RM, Casa DJ, McDermott BP, Ganio MS, Armstrong LE, Maresh CM (2009). "Does creatine supplementation hinder exercise heat tolerance or hydration status? A systematic review with meta-analyses". Journal of Athletic Training 44 (2): 215–23. doi:10.4085/1062-6050-44.2.215. PMID 19295968.
- ↑ 9.0 9.1 Dalbo VJ, Roberts MD, Stout JR, Kerksick CM (July 2008). "Putting to rest the myth of creatine supplementation leading to muscle cramps and dehydration". British Journal of Sports Medicine 42 (7): 567–73. doi:10.1136/bjsm.2007.042473. PMID 18184753.
- ↑ Poortmans JR, Francaux M (September 2000). "Adverse effects of creatine supplementation: fact or fiction?". Sports Medicine 30 (3): 155–70. doi:10.2165/00007256-200030030-00002. PMID 10999421.
- ↑ Francaux M, Poortmans JR (December 2006). "Side effects of creatine supplementation in athletes". International Journal of Sports Physiology and Performance 1 (4): 311–23. PMID 19124889.
- ↑ Bizzarini E, De Angelis L (December 2004). "Is the use of oral creatine supplementation safe?". The Journal of Sports Medicine and Physical Fitness 44 (4): 411–6. PMID 15758854.
- ↑ Bemben MG, Lamont HS (2005). "Creatine supplementation and exercise performance: recent findings". Sports Medicine 35 (2): 107–25. PMID 15707376.
- ↑ Kreider RB (February 2003). "Effects of creatine supplementation on performance and training adaptations". Molecular and Cellular Biochemistry 244 (1-2): 89–94. doi:10.1023/A:1022465203458. PMID 12701815. http://www.kluweronline.com/art.pdf?issn=0300-8177&volume=244&page=89.
- ↑ "How does Creatine stimulate muscle growth?". Creatine.com. 2007-03-05. http://www.creatine.com/en/news/2007/03/05/guarana-extract/#more-16. Retrieved 2010-08-16.
- ↑ Kamber M, Koster M, Kreis R, Walker G, Boesch C, Hoppeler H. Creatine supplementation--part I: performance, clinical chemistry, and muscle volume. Med. Sci. Sports Exer. 31: 1763-1769, 1999.
- ↑ Deldicque L, Décombaz J, Zbinden Foncea H, Vuichoud J, Poortmans JR, Francaux M. Kinetics of creatine ingested as a food ingredient. Eur. J. Appl. Physiol. 102: 133-143, 2008.
- ↑ R. Baselt, Disposition of Toxic Drugs and Chemicals in Man, 8th edition, Biomedical Publications, Foster City, CA, 2008, pp. 366-368.
- ↑ Hülsemann J, Manz F, Wember T, Schöch G (1987). "[Administration of creatine and creatinine with breast milk and infant milk preparations]" (in German). Klinische Pädiatrie 199 (4): 292–5. doi:10.1055/s-2008-1026805. PMID 3657037.
- ↑ Tarnopolsky M, Martin J (March 1999). "Creatine monohydrate increases strength in patients with neuromuscular disease". Neurology 52 (4): 854–7. PMID 10078740. http://www.neurology.org/cgi/pmidlookup?view=long&pmid=10078740.
- ↑ Klivenyi P, Ferrante RJ, Matthews RT, et al. (March 1999). "Neuroprotective effects of creatine in a transgenic animal model of amyotrophic lateral sclerosis". Nature Medicine 5 (3): 347–50. doi:10.1038/6568. PMID 10086395.
- ↑ Andreassen OA, Dedeoglu A, Ferrante RJ, et al. (June 2001). "Creatine increase survival and delays motor symptoms in a transgenic animal model of Huntington's disease". Neurobiology of Disease 8 (3): 479–91. doi:10.1006/nbdi.2001.0406. PMID 11447996.
- ↑ Rae C, Digney AL, McEwan SR, Bates TC (October 2003). "Oral creatine monohydrate supplementation improves brain performance: a double-blind, placebo-controlled, cross-over trial". Proceedings. Biological Sciences / the Royal Society 270 (1529): 2147–50. doi:10.1098/rspb.2003.2492. PMID 14561278.
- ↑ McMorris T, Mielcarz G, Harris RC, Swain JP, Howard A (September 2007). "Creatine supplementation and cognitive performance in elderly individuals". Neuropsychology, Development, and Cognition. Section B, Aging, Neuropsychology and Cognition 14 (5): 517–28. doi:10.1080/13825580600788100. PMID 17828627.
- ↑ Rawson ES, Lieberman HR, Walsh TM, Zuber SM, Harhart JM, Matthews TC (September 2008). "Creatine supplementation does not improve cognitive function in young adults". Physiology & Behavior 95 (1-2): 130–4. doi:10.1016/j.physbeh.2008.05.009. PMID 18579168.
External links
- NCBI Online Mendelian Inheritance In MAN (OMIM) GATM human mutation record
- Quackwatch on creatine
- Study on Safety of Creatine for Supplementation, 2010
- Creatine side effects: Fact or fiction?, 2010
- Creatine 'boosts brain power', BBC News, 12 August 2003
- Creatine: From Muscle to Brain, Peter W. Schutz, The Science Creative Quarterly, 2009
- Creatine during Pregnancy and Protection of Babies against Anoxia A mouse study
|
||||||||||||||